Electrochemical Electrode Developed to Generate Electricity from Hydrogen... Production Output Up 1.5 Times
- Input
- 2026-05-03 09:00:00
- Updated
- 2026-05-03 09:00:00
A research team led by Professor Seungho Cho of the Department of Materials Science and Engineering at Ulsan National Institute of Science and Technology (UNIST), working with Professor Jihwan An of Pohang University of Science and Technology (POSTECH), Professor Jeong Woo Han of Seoul National University, and Professor Bu Wenfei of Nanjing University of Information Science and Technology in China, said on the 3rd that it had developed a Solid Oxide Cell (SOC) electrode using layered double hydroxide (LDH) that does not lose performance even at high temperatures.
A Solid Oxide Fuel Cell (SOFC) is a device that generates electricity using fuels such as hydrogen or methane. Unlike the fuel cell used in hydrogen fuel cell vehicles (FCEVs), it can reverse the reaction when electricity is supplied. That means surplus overnight power can be used to produce hydrogen again, or carbon dioxide, a greenhouse gas, can be broken down to make carbon monoxide, an industrial gas.
The electrode developed by the team is highly durable because both the support and the catalyst are made of metal. When the new electrode was applied, it achieved a maximum output of 1.57 W/cm2 at 800 C when hydrogen was used as fuel, about 1.5 times higher than conventional electrodes. In another experiment, it also demonstrated durability by operating stably for 200 hours while producing carbon monoxide through the electrochemical reduction of carbon dioxide.
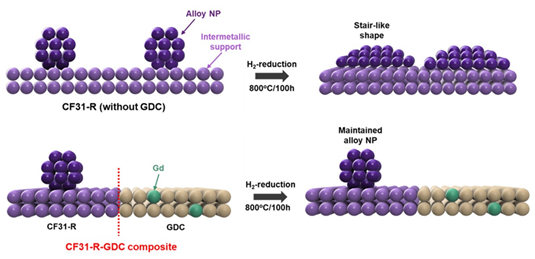
The reason the team was able to create an electrode with both a metal support and a metal catalyst lies in the raw material, layered double hydroxide (LDH). LDH is a material in which different metal ions are evenly mixed within a single layer, and those layers are stacked one on top of another. The process begins by heating an LDH containing cobalt and iron ions in air to harden a metal alloy framework that serves as the support. When it is heated again in hydrogen, alloy nanoparticles that act as the catalyst rise to the surface of the support through leaching.
The researchers were able to synthesize the material by systematically analyzing the internal structure, or phase transition, of LDH under different temperatures and heating conditions. They also added the additive GDC to the electrode to ensure a rapid supply of oxygen needed for the reaction.
The joint research team explained, "By reducing the need to replace electrodes and lowering operating costs, this could help popularize Solid Oxide Fuel Cells," adding that it is "a foundational technology that connects hydrogen and electricity production with carbon dioxide upcycling."
The study included first authors Kim Hyun-min, a graduate of UNIST's Department of Energy and Chemical Engineering and now at Stanford University; Kim Yun-seo, a graduate of UNIST's Department of Materials Science and Engineering and now at the Korea Institute of Science and Technology (KIST); and Seo Hwa-kyung, a researcher in the Department of Materials Science and Engineering at Seoul National University. The findings were published on April 16 in Advanced Functional Materials, a leading global journal in materials science.
jiany@fnnews.com Yeon Ji-an Reporter