"No Tariff Concerns" as K-Bio Targets U.S. Market on Patent Expirations and Wider FDA Designations
- Input
- 2026-03-02 15:03:19
- Updated
- 2026-03-02 15:03:19
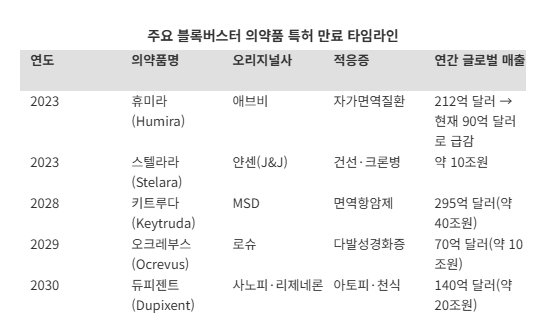
■ Rapid market share growth in the U.S.; first-mover advantage expectedAccording to industry sources on the 2nd, Celltrion's autoimmune disease treatment Yuflyma, a Humira biosimilar, posted 44% year-on-year growth in the United States in January and lifted its market share to 4.5%. The Adalimumab market, to which Yuflyma belongs, is worth about 10 trillion won. As AbbVie's original drug Humira has gone off patent, the switch to biosimilars is now in full swing.
In particular, Celltrion received two additional pediatric indications from the U.S. Food and Drug Administration (FDA) last year, for pediatric uveitis and Hidradenitis Suppurativa. The broader range of eligible patients has laid the groundwork for strong growth.
Beyond Yuflyma, Celltrion's other biosimilars are also maintaining solid competitiveness in the U.S. market. Inflectra, its Remicade biosimilar, holds about a 30% share by prescription volume and remains the market leader in its segment. Truxima, a biosimilar of Rituxan, has also achieved market shares in the 30% range in both the United States and Europe, growing 17.1% year-on-year.
Samsung Bioepis is likewise strengthening its presence in the United States. RENFLEXIS, its Remicade biosimilar, has surpassed a 10.4% share of the U.S. market, while HADLIMA, its Humira biosimilar, saw prescription volumes surge 52% from a year earlier, solidifying its position among the market leaders.
An industry official said, "Celltrion and Samsung Bioepis are already rapidly expanding their market shares in the United States," adding, "From this year, exemptions from Phase 3 clinical trials by the FDA are expected, which should further speed up market entry for new pipelines."
■ 600 trillion won patent cliff: competing through production bases and strategic operationsIn this context, patents on around 200 original drugs are scheduled to expire sequentially by 2030. The combined market size of these products is estimated to reach as much as 600 trillion won.
Once patents expire, biosimilars with strong price competitiveness can enter the market, and companies that secure products early are expected to expand their market shares quickly. Unlike in the past, when they were seen as latecomers, these firms can now operate their pipelines strategically in line with patent expiry schedules.
Their commercialization capabilities are also visibly strengthening. Late last year, Celltrion completed the acquisition of a manufacturing plant from U.S. drugmaker Eli Lilly and began full-scale production of Eli Lilly contract manufacturing organization (CMO) products across all lines in February. By securing a local production base, the company aims to enhance both supply stability and price competitiveness.
Samsung Bioepis is also pursuing multiple biosimilar approvals in the U.S. market as it seeks to expand direct sales. The company is moving beyond simply obtaining product approvals and shifting to a full value chain strategy that covers distribution, marketing, and insurance reimbursement.
An industry official noted, "The patent cliff is not just a temporary gap in the market, but a reshaping phase in which companies that have earned trust secure long-term market share," and went on to explain, "Since Celltrion and Samsung Bioepis have already built a strong track record, the next five years could be a period when K-Bio's global standing rises to a new level."
wonder@fnnews.com Jung Sang-hee and Kang Jung-mo Reporter