ABL Bio "Accelerating Clinical Trials Beyond 4 Trillion Technology Exports"
- Input
- 2025-07-28 15:39:50
- Updated
- 2025-07-28 15:39:50
Focusing on Achievements from BBB Shuttle to ABL111
Not Just Technology Export, Aiming to Lead the Cancer Market
[Financial News] The bispecific antibody specialist ABL Bio has announced its next-generation vision 'ABL Vision 2.0', unveiling a strategy to strengthen global competitiveness centered on the blood-brain barrier (BBB) shuttle platform 'Grabody-B' and the bispecific antibody 'ABL111'. Based on technology transfer achievements with global pharmaceutical companies and clinical data acquisition, the company expects its corporate value to surge after this year.Not Just Technology Export, Aiming to Lead the Cancer Market
On the 28th, ABL Bio held an online corporate briefing via YouTube, announcing the current status of major pipeline developments and mid-to-long-term strategies utilizing Grabody-T (4-1BB based immuno-oncology platform) and Grabody-B (BBB shuttle platform).
Sang-Hoon Lee, CEO of ABL Bio, stated, “Grabody-B has expanded its application range from existing antibody-centered to RNA-based drugs such as 'siRNA' and 'ASO',” adding, “We have also secured the possibility of drug delivery to muscle tissues, not just the brain.” ABL Bio confirmed the muscle targeting potential of Grabody-B through joint research with 'Ionis', and the related paper is expected to be submitted in August.
ABL Bio is collaborating with GSK, which has signed a 4 trillion won technology transfer contract for Grabody-B, based on new target foundations. They are devising a CNS (central nervous system) treatment strategy that excludes existing amyloid and tau, aiming for a safer and more efficient approach.
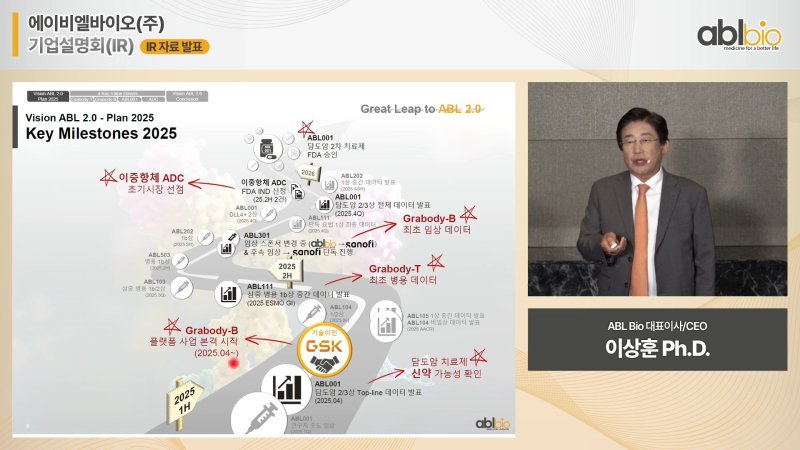
The pipeline that received the most attention at the conference was the bispecific antibody ABL111 (Zibastomig), which targets both Claudin18.2 and 4-1BB, expressed in gastric and pancreatic cancer patients. ABL111 showed encouraging results with an objective response rate (ORR) of 70.6% and a disease control rate (DCR) of 100% in Phase 1 and 2 clinical trials. In the high-dose group, the ORR reached 83%.
ABL Bio also announced that ABL001 (Tobesimig), being developed as a second-line treatment for bile duct cancer, is drawing attention in its Phase 2 and 3 clinical trials. ABL001 has been transferred to Compass Therapeutics in the United States.
CEO Lee stated, “We plan to announce progression-free survival (PFS) and overall survival (OS) data by the end of the year and expect accelerated approval from the FDA,” adding, “2025 will be the first year of significant corporate value increase, with the announcement of PFS data, FDA approval visibility, and expansion of platform partners.”
vrdw88@fnnews.com Kang Jung-mo Reporter